| Health / Health News |
Researchers unlock regenerative potential of cells in the mouse retina
Cells within an injured mouse eye can be coaxed into regenerating neurons and those new neurons appear to integrate themselves into the eye’s circuitry, new research shows. The findings potentially open the door to new treatments for eye trauma and retinal disease.
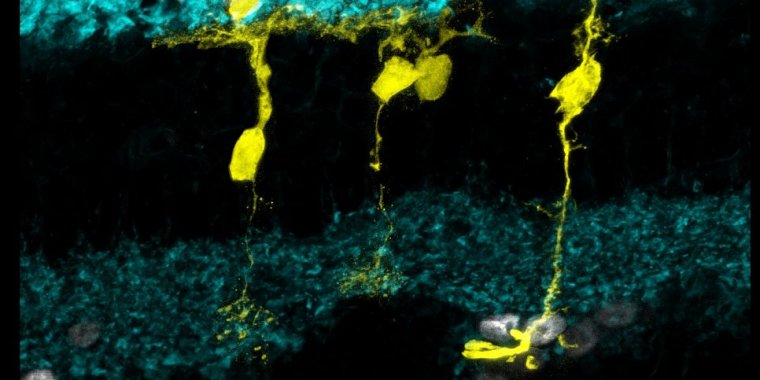
Regenerating Muller glia (yellow) in the mouse retina. ![]()
The study’s lead investigator, Tom Reh, Ph.D., and his team at UW Medicine in Seattle, looked to the zebrafish for clues about how to encourage regeneration in the mouse eye. When a zebrafish injures its eye, cells within the eye naturally regenerate, allowing the fish to maintain vision. Mammals lack this regenerative ability.
In studying zebrafish the research team homed in on Müller glia, a type of retinal cell that supports the health and functioning of neighboring neurons, and that also exhibits an innate regenerative ability. Sometimes referred to as the stem cells of the zebrafish eye, Müller glia are the cells from which all other types of retinal cells are regenerated in the fish.
Earlier research from Reh’s lab showed that in newborn mice, Müller glia can be directed to become retinal neurons by activating a transcription factor called Ascl1, which in turn activates a suite of genes involved in regeneration.
By the time the mice reach adulthood, however, regions of the genetic code that are targeted by Ascl1 and that are required for regeneration become inaccessible. In other words, in adult mice, regions of the genetic code that are critical for regeneration are closed for business.
They found that the commonly used anti-cancer agent trichostatin A (TSA) made critical regions of DNA accessible again. Ascl1 could then bind to those regions, which stimulated the regeneration of neurons in the adult mice.
The researchers injured the mice retinas with a toxin that causes cell death in retinal ganglion cells and interneurons, another type of retinal cell whose job it is to transmit signals from photoreceptors to the brain.
They then injected the mice with TSA and tamoxifen. Over the next several weeks, the shape and behavior of the fluorescent green-labeled cells were observed to see if there was evidence of regeneration.
Proteins expressed by the observed cells were similar to those of interneurons. Analyses of genome structure further shored up evidence that the cells that were once Müller glia had been genetically reprogrammed and were now showing characteristics of interneurons.
The studied cells had formed functioning synapses – connections from one neuron to another – and responded to light in a way that’s typical of a type of interneuron. The cells had also integrated with retinal cells that convey signals to the brain.
The findings are significant because they suggest the feasibility of a novel approach for encouraging regeneration in the mammalian retina, the light sensitive tissue at the back of the eye that dies in many blinding diseases. (National Institutes of Health)
YOU MAY ALSO LIKE




